US commission paves way for anthrax vaccine testing on children
Published 20 Mar, 2013 10:14 | Updated 20 Mar, 2013 10:40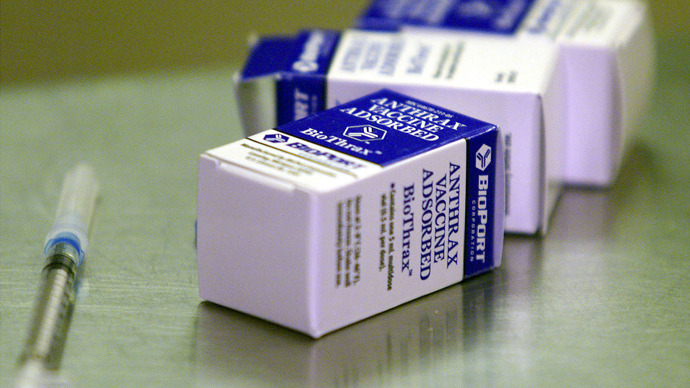
A US commission has advised that anthrax vaccination trials be carried out on children. The board gave strict guidelines for testing to put infants at no more than “minimal risk,” but critics accuse them of using children as guinea pigs.
The presidential commission described the necessity to test the
vaccine on children because in the event of a mass bioterrorist
attack a large proportion of the victims would be children. It
advised to start the testing of the vaccine on young adults aged
from 18 to 25 to first understand the risk before progressing on to
younger participants.
"You'd work your way down from 18-year-olds," said Dr. John
Parker, a retired army major general and chairman of the biodefense
board. "If it were safe you'd go to 17-year-olds, then 16-year
olds."
The commission, however, emphasized children would be subjected to
“no more than minimal risk” by the tests, despite strong
criticism from rights activists.
Health and Human Services (HHS) Secretary Kathleen Sebelius called
on the presidential commission to broach the ethical validity of
using children in vaccination testing.
The Commission Chair, Amy Gutmann, said that "this is one of the
most difficult ethical decisions the bioethics commission has
undertaken."
“Our nation ethically must protect children enrolled in research
studies, and we must also do our best to develop the knowledge
needed to save children's lives during a possible emergency,”
said Gutmann at a news conference on Tuesday.
Opposition to the report by the presidential commission likened
testing the vaccine on children to using infants as guinea pigs as
they are unable to fully understand the risks of participating in
such a study.
Vera Sharav, founder of the Alliance for Human Research Protection,
said that if testing on children were to be carried out it would
spell "moral harm for us as a nation and suffering for the
children. They should have said, 'thou shalt not’.”
Opponents to testing argue that antibiotics would offer sufficient
protection to children.
Side effects
The anthrax vaccine has been tested on around 2.9 million adults so
far, most of whom were members of the armed forces being vaccinated
against the threat of biological warfare in Iraq. A number of side
effects were registered during the testing of the vaccination,
including skin ulcers, fever and malaise.
"We have to wonder if, after all the data collected by the US
Army on the side-effects experienced by soldiers, we would want to
subject children to skin ulcers symptoms of the disease," said
Jeanne Guillemin, author of ‘American Anthrax’, to Reuters.
Additionally, a study carried out by the US Centers for Disease
Control and Prevention carried out a study in 2008 involving 1,563
adults who took the vaccine. Participants reported over 229 serious
adverse effects, such as seizures, intracranial aneurism and
cardiovascular disease, but only nine of these cases were
attributed to the vaccine.