Feds probe deaths linked to weight-loss balloons
Published 11 Aug, 2017 20:48 | Updated 13 Aug, 2017 06:34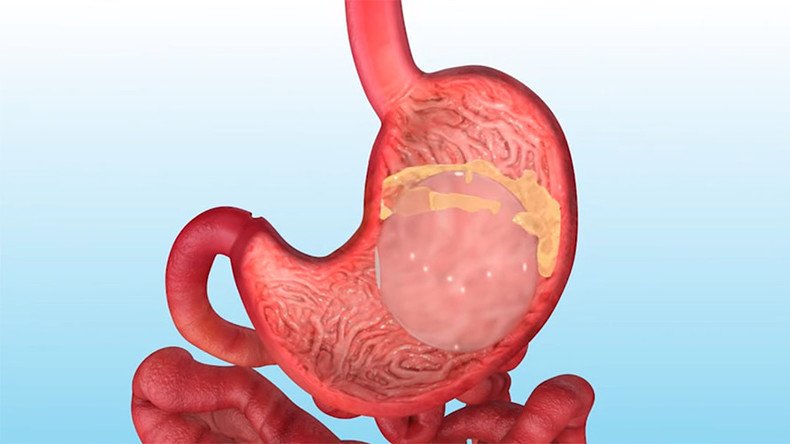
Weight-loss balloons have caught the attention of federal authorities in the US, after five people died within a month of being fitted with the devices.
A report by the US Food and Drug Administration (FDA) released Thursday states that the five people had undergone a procedure to be fitted with the saline-filled balloon since April last year. In each case, the patient died within a month or less. In three cases, the patients died within three days of the procedure.
The cause of the patients’ deaths remains unknown, the FDA noted in its report.
"At this time, we do not know the root cause or incidence rate of patient death, nor have we been able to definitively attribute the deaths to the devices or the insertion procedures for these devices," the report says.
'I’m saying officially right now it is an emergency. It’s a national emergency' - #Trump on #opioidshttps://t.co/DKKopryM0n
— RT America (@RT_America) August 10, 2017
In the procedure, a deflated silicone balloon is attached to a catheter and inserted through the throat into the stomach. Once the balloon hits the stomach wall, saline is pumped through the catheter and into the balloon. As a result, space is taken up in the stomach, making a person feel full after smaller meals, thereby decreasing their appetite.
Four of the deaths occurred after patients were fitted with the Orbera Intragastric Balloon System, which is manufactured by Apollo Endo Surgery. In one case, the person received a ReShape Integrated Dual Balloon System by ReShape Medical Inc.
British govt ‘squandering’ £1.6bn on ‘futile’ war on drugs - report. https://t.co/po6HMRqhhG
— RT UK (@RTUKnews) August 8, 2017
“The FDA’s letter to health care providers does not indicate that the patient deaths were related to the Orbera device or the insertion procedures,” Apollo Endo Surgery said in statement.
“While the cause of death has not been provided or determined in all cases, Apollo has not received any communication or indication from the attending physicians or hospitals that the deaths have been due to the Orbera device.”
There have been 21 reported deaths of people fitted with Orbera balloons since 2006, a rate of less than 0.01 percent of the 277,000 patients who have undergone the treatment, the company added.
While the investigation continues, the FDA has issued a recommendation that healthcare providers continue to closely monitor patients treated with these weight-loss devices.