Schizophrenia gene identified in landmark study, opening door to revolutionary treatments
Published 28 Jan, 2016 11:43
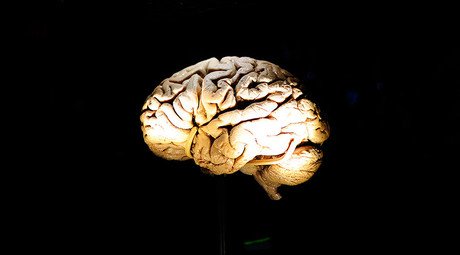
A startling discovery in the field of schizophrenia was made – scientists have pinpointed the brain mechanism that triggers it. The monumental study purports to reveal the very processes happening neurologically inside a sufferer’s head.
It is also the first study that conclusively ties schizophrenia to specific gene variants and a particular biological process, as well as helping explain decades of prior observations.
“Since schizophrenia was first described over a century ago, its underlying biology has been a black box, in part because it has been virtually impossible to model the disorder in cells or animals,” said senior author Steven McCarroll, from the Broad Institute of MIT and Harvard.
“The human genome is providing a powerful new way in to this disease. Understanding these genetic effects on risk is a way of prying open that black box, peering inside, and starting to see actual biological mechanisms.”
READ MORE: It’s all in the genes: Healthy people more likely to be intelligent, study says
The study, conducted by researchers primarily from Harvard, the Broad Institute and Boston Children’s Hospital, also opens doors to novel methods of early detection physicians thought were impossible just years ago, said Steven Hyman, the director of the Stanley Center for Psychiatric Research at the Broad Institute at MIT.
Hyman, who is also a former director of the National Institute of Mental Health, called the new research “the most significant mechanistic study about schizophrenia ever.”
The teams undertook the task of testing 65,000 people for the study, while gathering DNA from some 100,000, coming from 30 different countries. Complex analyses of genetic variation were carried out, with scientists finally arriving at chromosome 6, in a region of DNA long associated with infectious disease. This observation led to some proposing that schizophrenia itself could be triggered by an infectious agent. But until the study, no one knew how, since there were hundreds of genes in the region.
That is what led them to a specific region containing the gene called complement component 4(C4) – a well-known gene with regard to its role in the immune system and brain development. They were fortunate that C4 acts and appears differently in many individuals. This allowed for measuring variability.
This consequently led to scientists also tying it to the risk of schizophrenia. The particular structural form of the gene that leads to a higher expression of it in the immune system was almost always more prominently expressed in individuals with higher risk of schizophrenia. Knowing this greatly increases chances of new therapy and preventative measures, they said.
They discovered that the risk of schizophrenia increases dramatically in people who inherit a particular gene connected to “synaptic pruning” – a process severing connections between neurons. Now that we know the biological origins of schizophrenia to a much greater degree, the doctors look toward research on how to reduce synaptic pruning in individuals showing early symptoms.
"This study marks a crucial turning point in the fight against mental illness," said Bruce Cuthbert, acting director of the National Institute of Mental Health, who was not involved in the research. "Because the molecular origins of psychiatric diseases are little-understood, efforts by pharmaceutical companies to pursue new therapeutics are few and far between. This study changes the game. Thanks to this genetic breakthrough we can finally see the potential for clinical tests, early detection, new treatments, and even prevention."
READ MORE: Increasingly twisted sense of humor could be early sign of dementia – study
The study appears in the latest issue of the journal Nature.
Approximately 1 percent of the American population is estimated to be afflicted with the debilitating condition that strikes at the core of a person’s cognitive functioning. Until this point, no effective treatment for the condition has been offered by medicine. The new approach to targeting complementing proteins fills scientists with hope for a whole range of treatment down the road.