Life-forming molecule discovered in deep space may hold key to life on Earth
Published 16 Jun, 2016 01:33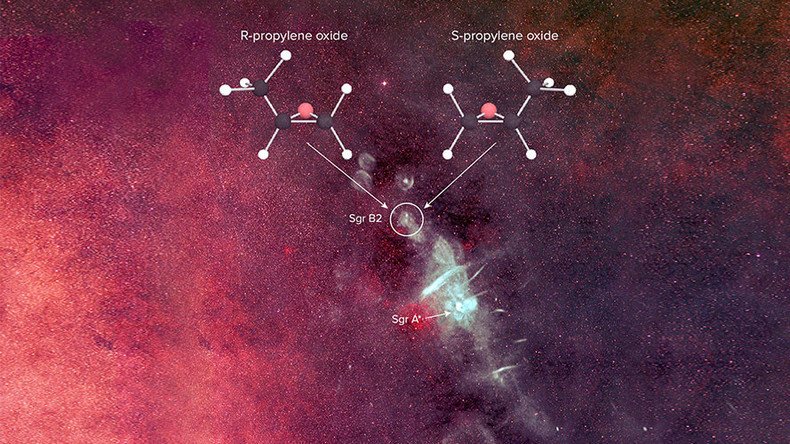
Scientists are one step closer to proving we’re all made of stars with the discovery of a complex life-forming molecule in deep space. A chiral molecule essential for life was detected in a giant gas cloud 390 light years away from the Milky Way’s center.
The National Science Foundation’s Green Bank Telescope in West Virginia, working alongside the CSIRO’s Parkes telescope in Australia, detected the molecule known as propylene oxide in the solar cloud Sagittarius B2.
Chiral molecules, previously undetected in deep space, have baffled scientists trying to determine how they form life for years.
“This is the first molecule detected in interstellar space that has the property of chirality, making it a pioneering leap forward in our understanding of how prebiotic molecules are made in the Universe and the effects they may have on the origins of life,” Brett McGuire, a chemist and astronomer involved in the research told Science Daily.
Chiral molecules employ what is known as homochirality, also called one handedness, meaning they form to either the right or to the left, but never both. This feature is key to DNA’s double helix.
Just why they go to one side and not the other has puzzled scientists for years, but the new discovery may help them find the answer. Brandon Carroll, a chemistry graduate and researcher for the study, told Science Daily that the molecules may form naturally in space before being deposited on young planets via asteroids and comets.
“Meteorites in our Solar System contain chiral molecules that predate the Earth itself, and chiral molecules have recently been discovered in comets,” according to Carroll.
“It is strong evidence that life on earth really began, in its earliest forms, by these molecules being produced in deep space, around star forming regions,” Dr. John Reynolds from CSIRO said.
The universe is (sometimes) a lefty: 1st detection of chiral molecules in the ISM https://t.co/4oMhaMxpXD#astronomypic.twitter.com/PWyAkSz15p
— Dr. John Barentine (@JohnBarentine) June 15, 2016
The story behind direct mass spectrometric detection of chiral molecules using MS-PECD https://t.co/QflpWG943xpic.twitter.com/TCWWsehGYF
— Analytical Scientist (@tAnaSci) June 14, 2016
The molecule was detected when satellites measured multiple spectral lines associated with propylene oxide. Data could not distinguish between the left and right handedness of the molecules, something the researchers hope to achieve eventually by examining how polarized light interacts with them in space.
The findings were published in the journal Science this week.











