Artificial wombs successfully pass 1st test, human trials could begin within 3yrs (PHOTOS)
Published 25 Apr, 2017 20:10 | Updated 25 Apr, 2017 22:15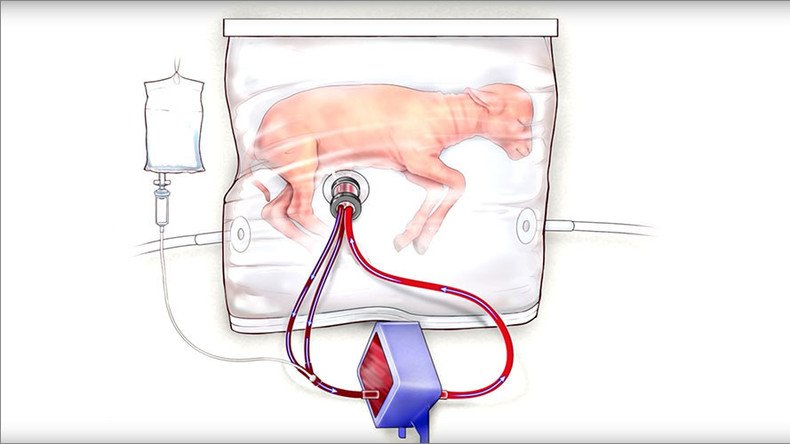
Scientists have successfully developed and tested an artificial womb capable of supporting prematurely born lambs for periods of up to four weeks in a landmark development that could dramatically reduce the risks posed by premature births in humans.
Lambs born at the equivalent point of 23 weeks into the human gestation period have been kept alive in a transparent vessel, or ‘biobag,’ that serves as both a womb and incubator for periods of up to four weeks after their initial, premature birth.
The research was conducted by a team of doctors and scientists led by Alan Flake, a fetal surgeon at the Children’s Hospital of Philadelphia. The team’s findings were published in the journal Nature on Tuesday.
“If we can support growth and organ maturation for only a few weeks, we can dramatically improve outcomes for extremely premature babies,” said Flake when speaking to the media.
First heartbeat of babies as early as 16 days after conception - study https://t.co/193xQxLSNEpic.twitter.com/RJeCqzSlDp
— RT (@RT_com) October 13, 2016
The ‘biobag’ device consists of three main parts: A pumpless arteriovenous circuit, which keeps the fetus’ blood flowing but relies on the infant’s heart to do the work so that it develops properly; the closed, dark and sterile fluid environment that protects the barely developed skin organ from infection and damage; and a unique artificial system of umbilical vascular access that emulates the variety of functions performed by a mother’s umbilical cord.
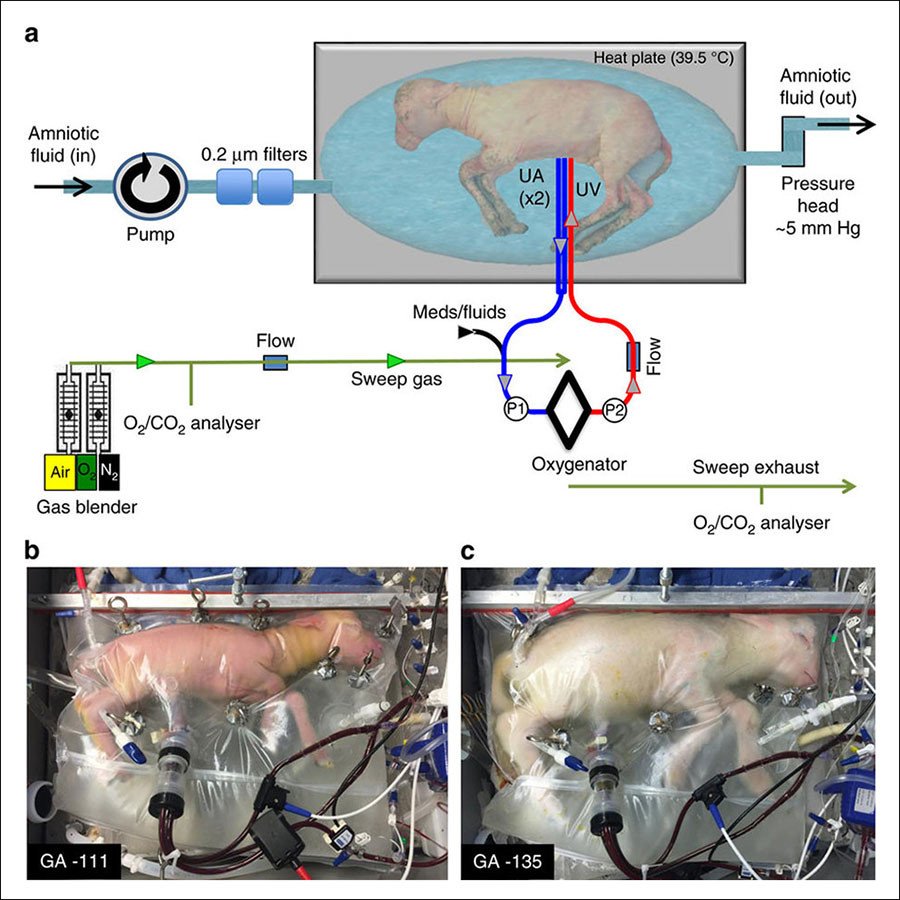
(a) Circuit and system components consisting of a pumpless, low-resistance oxygenator circuit, a closed fluid environment with continuous fluid exchange and an umbilical vascular interface. (b) Representative lamb cannulated at 107 days of gestation and on day 4 of support. (c) The same lamb on day 28 of support illustrating somatic growth and maturation.
These three devices effectively mimic the role performed by a mother’s placenta, providing clean, oxygenated blood and nutrients to the fetus, while also removing waste matter to prevent toxicity.
While it’s too soon in the technology’s development to entirely replace a mother, or surrogate’s, womb, medical professionals believe artificial wombs could serve as a novel, life-saving solution for human babies born between 23 and 28 weeks gestation.
Three-parent baby could be born this year after new fertility technique gets go-ahead https://t.co/xpGeUhQVIJ
— RT (@RT_com) March 16, 2017
Babies born prematurely at this critical stage of development cannot breathe, feed or fight off infection without medical assistance, and even with artificial life support, often suffer from debilitating conditions which greatly impact their quality of life, and mortality rates.
According to the researchers, over one-third of all infant deaths and 50 percent of cerebral palsy cases can be attributed to premature births.
Of those fetuses that do survive, many suffer from chronic lung disease and other associated complications stemming from organ immaturity.
Flake was quick to point out that we won’t be seeing test-tube or bag-grown babies like the ones described in Aldous Huxley’s A Brave New World anytime soon, however.
“I don’t want this to be visualised as humans hanging on the walls in bags,” said Flake. “This is not how this device will work or look.”
“The reality is that at the present time there’s no technology [like that] on the horizon… There’s nothing but the mother that’s able to support that [initial] period of time,” he said.
Flake and his team expect that, pending FDA approval, human clinical trials could begin in the next three years.












