Motherless babies? Egg fertilization not needed for reproduction, scientists say
Published 14 Sep, 2016 10:12 | Updated 20 Sep, 2016 14:11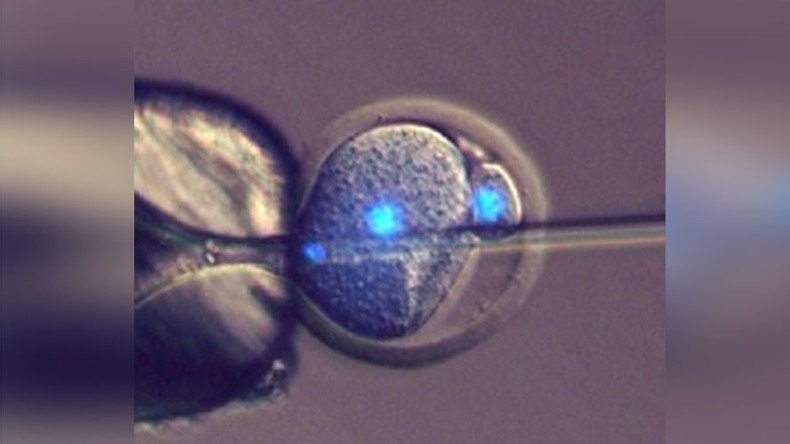
Motherless babies could be a real possibility in the near future, according to scientists who managed to create a litter of mice without fertilizing a female egg. The breakthrough could help infertile women have children.
The old-fashioned way of making a baby is quite simple – sperm fertilizes an egg, and a baby is born nine months later. But scientists from the University of Bath are questioning the necessity of an egg in the equation, after successfully producing a litter of mice by bypassing the fertilization process.
The trick lies within something called parthenogenotes – egg cells that have been “tricked” into becoming embryos without being fertilized by sperm.
However, scientists had to come up with a way to keep the parthenogenotes alive, as they die by themselves after just a few days. It turned out injecting them with mouse sperm was the answer.
Instead of dying, the embryos developed normally. They were then transferred into female mice, and became perfectly healthy mouse pups.
In total, the researchers produced 30 pups, with a success rate up to 24 percent. Some of those pups are now grand-mice and great-grand-mice, as their offspring have gone on to have pups of their own.
"This is first time that full term development has been achieved by injecting sperm into embryos," Tony Perry, a researcher at the University of Bath, said in a news release. "It had been thought that only an egg cell was capable of reprogramming sperm to allow embryonic development to take place."
Perry went on to say that his team's work challenges “dogma held since early embryologists first observed mammalian eggs around 1827 and observed fertilization 50 years later, that only an egg cell fertilized with a sperm cell can result in a live mammalian birth."
The research, according to Perry, could open up doors for revolutionary ways to create embryos.
“What we’re talking about are different ways of making embryos. Imagine that you could take skin cells and make embryos from them? This would have all kinds of utility,” he said.
If such a process became available to the masses, it could help women who were infertile. It could also assist in the saving of endangered animal species.
Perry said he and his colleagues are aiming to further study the potential for skin cells to replace eggs.
“Will we be able to do that? I don’t know. But I think, if it is ever possible, one day in the distant future people will look back and say this is where it started.”
The study was published in the journal Nature Communications.