World's first biolimb: Scientists create living, functioning rat leg
Published 4 Jun, 2015 17:26 | Updated 6 Jun, 2015 07:53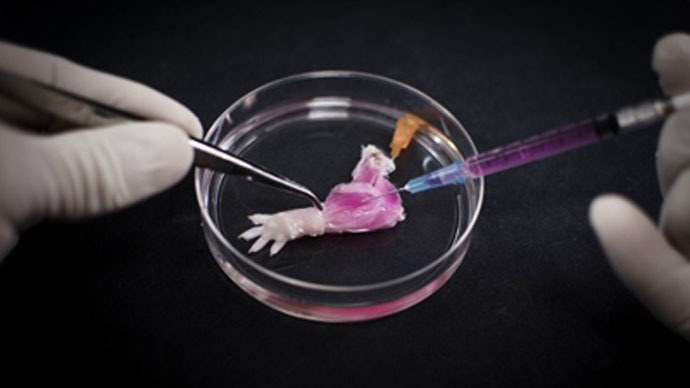
Scientists have created the world's first lab-grown biolimb – a rat leg that circulates blood and responds to stimuli. The breakthrough could lead to amputees one day growing their own replacement limbs.
The rat leg, grown by researchers at Massachusetts General Hospital, has functioning veins and muscle tissue – but the journey toward a living limb actually began with a dead rat.
That journey, led by Dr. Harold Ott for the MGH Department of Surgery and the Center for Regenerative Medicine, will be published in the journal Biomaterials in August.
First, Ott and his team stripped living cells from a dead rat, using a technique called decellularization. This involved soaking the limbs of the dead rat in a detergent solution for a week. The process exposed the rat's scaffolds, or non-living parts.
Meanwhile, vascular and muscle cells were grown in a culture. They were then planted onto the scaffolds in a process called recellularization.
The limb was placed inside a bioreactor which was filled with a nutrient solution to grow the cells. Electrical stimulation was applied after five days, in order to promote muscle formation.
It was removed from the bioreactor after two weeks. The result was a scientific breakthrough of no small scale: a real, living rat leg – minus the bones and cartilage.
The next step was to attach the leg to healthy rats, to determine whether the limb would actually function.
Once that was done, Ott found that the blood from the rats circulated to the new limb as intended. When stimulated, the leg's muscles also functioned with 80 percent strength of those in newborn animals.
The same technique has been used to create synthetic bladders, windpipes and other organs, though not always successfully. Those organs were also a lot simpler, as they had only a few types of cells and aren't expected to perform complex tasks. A limb, however, is much more intricate and difficult to replicate.
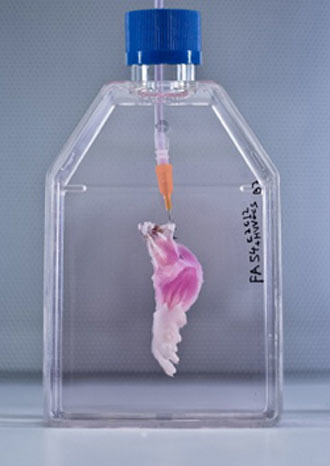
The pioneering breakthrough could be life-changing for amputees, potentially allowing them to one day grow their own replacement limbs.
This would come with a number of advantages over robotic prosthetics.
For starters, the person would not have to train their brain to control the limb. It would also be free of the limitations experienced with prosthetics, including the inability to gauge pressure or heat.
“Even with prosthetics, many patients who have forearm loss do not regain full independence in terms of taking care of themselves, because the dexterity of these devices is not quite there yet,” Ott said.
Limbs made from one's own cells would also trump those donated by other people, because the body would be much less likely to reject them.
Doctors have successfully transplanted donated hands and arms, but recipients have to take drugs that suppress their immune systems for the rest of their lives so their bodies don't reject them.
But the possibility of widespread human biolimbs is still in the distant future. Ott believes it will be at least a decade before the first ones are ready to be tested.
He also stressed that science shouldn't move too quickly, as patients' lives need to be protected.
“What you never want to do is put a patient at an unnecessary risk for a small benefit,” he said.
But Ott is optimistic about the future, believing that people will one day donate limbs like they currently donate kidneys. Those limbs will undergo the same process as the rat's leg – stripped of their soft tissue, planted with new cells, and given to amputees.












